Practical battery-back-up power for amateur radio stations 1
Many radio amateurs use batteries only in low-power, portable/ mobile applications. Here's how to use batteries to keep a ham station going when commercial ac power fails.
Heavy-duty back-up power is readily available for many - perhaps most - Amateur Radio stations, and it need not be expensive. Virtually all modern amateur gear is designed to operate from 11 to 14 or 15 V dc, making operation possible from highly reliable, heavy-duty batteries. Yet, battery backup is underappreciated and underutilized by amateurs - probably because of a lack of familiarity and the supposed difficulty or expense of obtaining suitable batteries. Surplus commercial or industrial heavy-duty lead-acid cells often can be obtained for the asking, however, and frequently they come in batches big enough to supply several stations at once.(1)
Besides superb back-up against failure of commercial ac power, batteries provide other benefits. One of these is surge protection: A power-line spike big enough to demolish a regulated dc power supply probably won't get past a battery to damage solid-state electronics. In addition, batteries:
- Are clean and safe.(2)
- Require very little maintenance.
- Offer considerable overvoltage protectiqp if a power-supply regulator fails.
- Can accumulate dribs and drabs of energy from alternative power systems, such as solar energy, water or wind power, and deliver it when needed.
As useful and versatile as batteries are, however, building a battery back-up system requires care and planning. Many hams believe that an old automobile battery, stuck under the operating desk and put on a charger every so often, constitutes battery back-up. 'Tain't so!
This three-part article describes how to get a practical battery-back-up system up and running. In this article, Part 1, we'll discuss back-up-battery types, chemistry, construction and procurement, and the basics of housing and installing a power-back-up system. In Part 2, we'll cover battery chargers and charging. Part 3 will cover back-up-battery monitoring, maintenance and safety, and how to dispose of unusable batteries safely and responsibly.
Components of a battery-back-up system
In its most basic form (Fig 1), a batterybacked-up power system consists of a backup battery, its load, a charger, and interconnecting wiring. A practical system - Fig 2 outlines mine - may include more than one storage battery, and also includes fusing and instruments (or points for connecting instruments) to monitor voltage and current in the system.
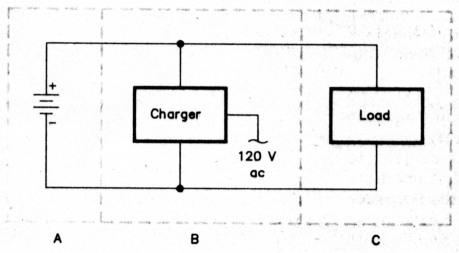
Fig 1 - A basic battery-based power system consists of a back-up battery, A, a charger, B, the system load, C, and interconnecting wiring. This article, part 1 of 3, focuses on back-up-battery topics and the basics of housing and installing a power-back-up system. Chargers and charging topics are discussed in Part 2.
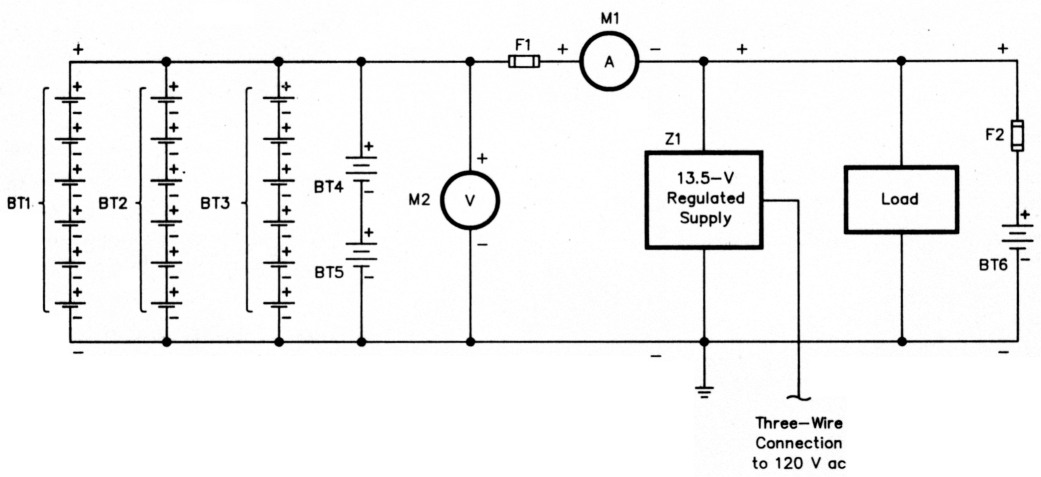
Fig 2 - A practical battery-backed-up power system may include more than one storage battery, and also includes fusing and instruments, or points for connecting instruments, to monitor system voltage and current. This drawing, which depicts the author's system, Illustrates how batteries of varying types and capacities can work together to provide solid back-up power. Stock regulated power supplies usually must be ritodftd before they can be used for charging service at Z1; see below and Part 2 of this article - to appear In a subsequent issue of QST - for more information. A simple battery voltage/current monitor (M1 and M2 serve this function in this diagram) will be described In Part 3.
BT1, BT2, BT3 - Battery consisting of six 2-V, 300-Ah float cells.
BT4, BT5 - 6-V, 100-Ah deep-cycle batteries.
BT8 - Optional deep-cycle battery to handle peak current demand in cases of high-voltage drop in station-to-battery wiring; see text.
F1, F2 - 32-V fuse of a current rating no greater than that necessary to handle the system current demand with a margin of safety (for example, 20 to 25 A for the 15-A load presented by a 13.8-V, 100-W MF/HF transceiver). F2 is used only if BT6 is present.
M1 - Zero-center ammeter; range dependent on battery capacity and load.
M2 - Meter capable of measuring voltages from 10 to 15. A 0 to 15-V meter will work, but a 10- to 15-V, expanded-scale meter is better.
Z1 - Regulated power supply, modified for protection against reverse voltage as described in Part 2 of this article. Do not use a supply that has not been modified for reverse-voltege protection.
Suitable batteries for back-up power
Automotive batteries are unsuitable for station back-up power except as a last resort.(3) Intended to start cars, they are designed to provide several hundred amperes for the few seconds needed to crank an engine. From a back-up-power standpoint, though, their weakness is that they don't like to be discharged very far. You may have already discovered this the hard way: Kill your car battery two or three times by leaving your headlights on, and you'll probably have to replace it.

Part of the author's 12-V back-up power system, these 2-V, 300-Ah, lead-acid float cells - obtained free of charge from a local telephone company - help keep W4MLE on the air when commercial power fails. (W4MLE photos)
Three other types of rechargeable lead-acid battery, readily available to radio amateurs, can be deeply discharged and recharged:
- Deep-cycle batteries are used in recreational vehicles and by boaters and fishermen to operate boat trolling motors, lights, pumps and electronics. Industrial deep-cycle batteries are often fully discharged every day and recharged every night.
- Gelled-electrolyte batteries, manufactured both for float and deep-cycle service, are usually intended for portable use and can be quite small. In most respects, they can be managed either as conventional deep-cycle batteries or, in light-duty applications, as float batteries. I won't discuss gelled-electrolyte batteries specifically because they behave much like their liquid-electrolyte counterparts.
- "Float-service" batteries, used in uninterruptible power systems(4) for telephone and large computer systems, should not be discharged and recharged more frequently than necessary. They are kept on a regulated charging source all the time - the same source that normally powers the load.
Properly applied, deep-cycle, gelled-electrolyte and float-service batteries are well-suited for back-up service even though their chemistries are similar to that of automotive batteries. They can replace your station's commercial ac power so completely during an outage that your rig may not notice that the power is down!
Back-up battery chemistry
Selection, use and care of lead-acid backup batteries requires knowledge of their chemistry and construction. Most such batteries consist of a case, or tank, divided into compartments - one compartment per cell - each of which is filled with dilute sulfuric acid (H2SO4). Immersed in this acid are plates - actually latticework frames - made of lead alloy. Half of the total number of a given cell's plates are filled with lead peroxide (PbO2); these plates serve as the cell anode (+ electrode). The remainder of the cell's plates are filled with spongy metallic lead (Pb) - lead that's finely divided to expose maximum surface area to the electrolyte. These spongy plates serve as the cell cathode (- electrode).
Deep-cycle cells generally differ in construction from automotive cells in that their plate-support grids consist of a lead-antimony alloy that stands up well to repeated cycling.
Float cells usually contain lead-calcium plates that tend to respond to repeated cycling by swelling, cracking and falling apart, eventually creating short circuits between adjacent plates.
With no load connected to a fully charged battery of any of these types, a voltage - about 2 V per cell - appears between the anode and cathode of each cell. When a load is connected, the battery releases stored chemical energy by causing electric current to flow through the load. The transformation of chemical to electrical energy occurs as a result of several chemical reactions in the battery, with the final results represented by the equation:
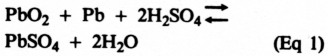
Translated, this means that the lead peroxide and spongy lead react with the sulfuric acid to release energy and produce two new compounds - lead sulfate (PbSO4) and water. This reaction is indicated by the right-pointing arrow in Eq 1. As the discharge continues, more and more acid is converted, causing the specific gravity of the electrolyte to decrease.(5) When all the lead peroxide (PbO2) is used up, the battery is "dead." Because the electrolyte normally contains a surplus of acid, its specific gravity usually doesn't fall all the way to 1.0.
When the battery is charged, the reaction reverses (hence the left-pointing arrow in Eq 1), converting the charging electrical energy into stored chemical energy. Now, lead sulfate on the positive plate is converted back to lead peroxide, releasing sulfuric acid. Lead sulfate on the negative plate is converted hack to spongy least, producing still more sulfuric acid. When all the sulfuric acid has been released, the battery is fully recharged and the specific gravity of the electrolyte is hack at maximum.
The chemical process is the same in lead-calcium and lead-antimony cells (including gelled-electrolyte cells). But the design and application of these two types differ in detail, such as the optimum electrolyte specific gravity, the normal open-circuit voltage for a single cell at full charge, and the optimum float voltage. Lead-calcium cells, usually used in float-charged systems, require and produce a slightly higher voltage per cell, lose less energy through internal leakage and take less maintenance, than their lead-antimony counterparts. These design differences are important in Amateur Radio applications.
Since back-up batteries are seldom, if ever, called upon to deliver short bursts of extremely high current, their electrolyte is not as acidic as that in automotive batteries, and they contain fewer plates.(6) On the other hand, back-up-battery plates are thicker and more densely packed with material than those of automotive starting batteries. And the plates of float batteries are usually immersed in larger quantities of electrolyte than other lead-acid battery types.

These 6-V, 100-Ah, float batteries, also part of the W4MLE back-up battery, consist of three cells each.
An overview of available back-up batteries
Float-service batteries are the vice presidents of the battery world. Their functions are to wait for a main power system to fail, to be ready when it does, and to not complain when it doesn't. They are electrochemical couch potatoes, luxuriating in a light but continuous flow of current - just enough to replace their internal leakage losses - from a regulated power supply.
Float cells are sold in many forms. They range from 6-V, 100-Ah batteries of three cells each, to individual 2-V, 100- to 300-Ah cells weighing as much as 85 pounds each. Cells of much higher capacities are manufactured for industrial uses, but their size usually precludes their use in ham stations.
Most manufacturers, such as Gould, Exide and C&D, warrant their float cells for 20 years under strenuous industrial use. Used for power back-up in Amateur Radio stations, industrial float batteries may be virtually immortal, with industrial preventive-maintenance discards probably lasting more than 10 years even after a hard working life. Remember, though, that float cells don't like deep discharges. One manufacturer(7) warns that if occasional discharges are experienced, battery life will decrease in proportion to the frequency and depth of these discharges.
It is customary that a stationary battery will not experience any more than 200 discharge cycles evenly distributed throughout its useful life. Frequent or greater depths of discharge can shorten service life to 10 years or even less, even with proper maintenance and operating conditions.
Two hundred cycles may sound like a lot, but remember that many industrial applications - golf-cart service, for instance - require full discharge every day and complete recharge every night. In such service, 200 cycles amounts to less than a year of business days!
Deep-cycle batteries can tolerate frequent discharges to about 11 V, and can withstand such use every day for years if well-maintained. But they also perform quite well in float service. The most useful deep-cycle batteries for Amateur Radio Service consist of six 2 V cells, connected in series, in a single, compact case. Deep-cycle batteries intended for use on boats and recreational vehicles are typically rated at 60 to 100 Ah, compared to 40 to 50 Ah for car (starting) batteries. And deep-cycle batteries are designed to operate at 13.8 V while charging. Industrial deep-cycle batteries may be designed for slightly lower voltages.
Deep-cycle fishing and recreational-vehicle batteries are normally warranted for 18 months because they tend to be abused by forgetful sportsmen who top them off by fast charging after leaving them discharged for long periods. Properly nurtured and used in stationary service at a ham station, however, deep-cycle fishing batteries can last much, much longer than 18 months.
Obtaining back-up batteries
Where can you find suitable back-up batteries? You could buy float cells from a manufacturer for something like $1/Ah per 2 V cell. That amounts to maybe $300 for a 2 V, 300 Ah cell $1,800 for a 6 cell battery! Surprisingly, however, float cells are often available gratis.
Used batteries are seldom retained when float-cell-backed power systems are upgraded. Old float cells are generally hauled off to a toxic-waste dump or to a salvage house, where the cells' lead, and polycarbonate plastic cases, are reclaimed. Often, an Amateur Radio club, or even individual amateurs, can arrange to get these discarded cells for hauling them away.
The Tallahassee Amateur Radio Society has a written agreement with the local telephone company (Centel) to accept discarded batteries on behalf of club members. About 75% of the cells the club has received from the company were in good health. Similar arrangements have been worked out between industrial battery users and other Amateur Radio clubs nationwide. My own station has a 1000 Ah floatcell bank, some cells of which once powered the city police department's telephones. Other cells powered a monster computer at Florida State University!
Of course, deep-cycle batteries can be purchased off-the-shelf at most boat dealers, fishing-tackle shops and automotive stores. Price, warranty and rated capacity are the important considerations in a new-battery purchase.

W4MLE's main battery - BT1-BT5 in Fig 2 - consists of deep-cycle and float cells, connected in series-parallel and housed outdoors.
Housing a battery-back-up system
Back-up batteries can be kept indoors, since they do not sputter electrolyte as automotive batteries do, but they must be ventilated. Charging generates oxygen and hydrogen - gases that form a highly explosive mixture if allowed to collect in an enclosed area. With back-up batteries, safe ventilation is generally assured if you follow a simple rule: Don't put them in a closet or enclose them in a box. Small batteries can be stored in a corner or under a desk; a room's ordinary air circulation will suffice for ventilation in most cases.
Back-up batteries can be kept outdoors in most climates; fully charged, they freeze at about -95°F.(8) Completely discharged - when their electrolyte is least acidic - their freezing point is quite close to that of water (32 °F). To protect the battery from sunlight, rain, snow, trash, insects and accidental short circuits, the cells should be provided with a housing or cover. You may be able to obtain a suitable plastic-coated steel battery rack from the supplier of your back-up cells. Such racks can usually hold two sets of six cells each. Or you can build a rack of your own, as I did the rack shown in the photo above. If you build your own rack, be sure it can take the weight of your back-up batteries; my rack consists of 2 x 4 and 4 x 4 lumber.
The porous, explosion-proof caps in the tops of back-up-batteries' cells are normally covered by plastic lids. If these lids get lost, don't leave the caps open; trash, rain and insects can get in. (Mud-dauber wasps, for instance, love to build nests in the necks of the funnels - and mud contains no known vitamins beneficial to battery cells.) Ordinary glass marbles, placed in the cap necks, are a good substitute for the lids; they can be easily removed when water must be added to the cells.
Back-up batteries need protection from direct sunlight. The polycarbonate plastic cases of float cells can be damaged by ultraviolet light; they develop cracks, and leak. If you store your cells outdoors, keep them off the ground and out of the sun, where you can eyeball them frequently to check their electrolyte levels. At nontropical latitudes in the northern hemisphere, the north side of a building receives little or no sun throughout the year; if that's where you live, the north side of your house is a good location for battery storage, when it's available.
Minimizing voltage drop in battery-to-station wiring
Wire is resistive, and resistance dissipates useful power as heat. Because of this, be sure to install your back-up battery as close as possible to its heaviest load - no more than 10 to 15 ft away, and the closer the better. A 200 W load, such as a 100 W output MF/HF transceiver, draws at least 15 A at 13.5 V; a 300 W load draws 22 A at this voltage. The wire resistance between battery and load causes a voltage drop; for example, a resistance of 0.1 Ω in a line carrying 22 A causes a drop of 2.2 V! The resistance of the system's positive and negative leads must be taken into account. Moral: Use heavy-gage wire.
Automotive battery cables are a good source of such wire. Welding cable is another. Or, you can try flexible, three-conductor, house-wiring cable: Double it and connect all six of its no. 10 solid conductors in parallel. Use one such six-conductor cable for the positive connection, and another for your system's negative loadto-battery lead. (You may be able to find some suitable discarded cable at a building site, or obtain "reel ends" from a building-materials supplier.) Make sure that every connection is well soldered and mechanically tight. You may have to use a propane torch to heat the joints sufficiently to solder them. Connect the main-battery negative lead to the power supply and the rig.
Assuming that the battery is sufficiently close to its load, most of the resistance in a back-up system occurs at its connectors. Measure the system voltage at the battery, at the power supply and at the load. If you measure a drop of more than half a volt at full load, you have a problem. (Monitors for in-line current and voltage at the battery terminals should be located where you'll see them every time you enter the room. Part 3 of this article will describe battery monitoring in detail and describe a circuit designed for this purpose.)
In cases of excessive voltage drop, such as those with long runs of wire (15 feet or more), another solution is possible. Put a small (100 Ah) battery under your operating desk, within two or three feet of the system's heaviest load (usually your MF/HF transceiver). Hang this additional battery BT6 in Fig 2 - across the 12 V line by connecting it directly - through a suitably rated fuse in its positive lead - to the rig terminals, in parallel with the line from the outside batteries and power supply. This battery will keep the system voltage from sagging during the relatively brief periods of heavy load and then recharge itself from the main battery during intervals of light load.
Notes
- A cell is a single electrochemical unit capable of generating electricity by means of two electrodes and an electrolyte. Depending on its chemistry and design, a cell may be classed as primary (non-rechargeable) or secondary (rechargeable). A battery consists of two or more cells connected in series, parallel or series-parallel to combine their voltage and/or current capacities. (In popular usage, battery can mean a single cell or a battery of cells. For clarity, battery is used to mean "a battery of cells" throughout this article.) The batteries used in commercial and Amateur Radio back-up service are invariably rechargeable and, in heavy-duty, full-station back-up service, are nearly always based on lead-acid chemistry. Thus, throughout this article, back-up battery is used to mean "lead-acid back-up battery."
- But reasonable safety precautions must be taken when handling and installing back-up batteries because they contain caustic and highly corrosive sulfuric acid.
- "Standby Power Will the Automotive Battery Do?" Telephone Journal, Jan-Feb 1975.
- Uninterruptible power systems are designed to be just that: power-supply systems capable of taking over, or continuing to operate, when regular ac power fails.
- The specific gravity of a substance is the ratio of its density to the density of another substance, with both densities measured in air. The specific gravity of back-up-battery electrolytes uses that of pure water - 1.0 - as a referent. The specific gravity of the electrolyte in fully charged float cells typically ranges from about 1.2 to 1.275; for deep-cycle cells, the specific gravity is slightly higher.
- Float-service cells usually contain approximately 6 molar sulfuric acid; this is, six moles (molecular weights, measured in grams) of H2SO4 per liter of electrolyte. The molecular weight of sulfuric acid is 98, so a mole of sulfuric acid is 98 grams.
- C&D Stationary Battery Installation and Operating Instructions, pub in 1981 by C&D Batteries, 3043 Walton Rd, Plymouth Meeting, PA 19462.
- If you store your batteries outside, be sure they are inaccessible to unauthorized personnel.
W4MLE, George L. Thurston III.